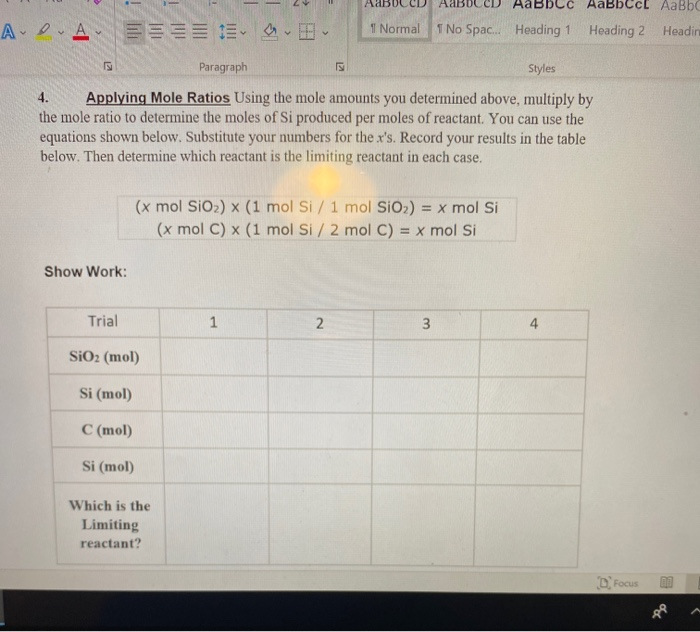
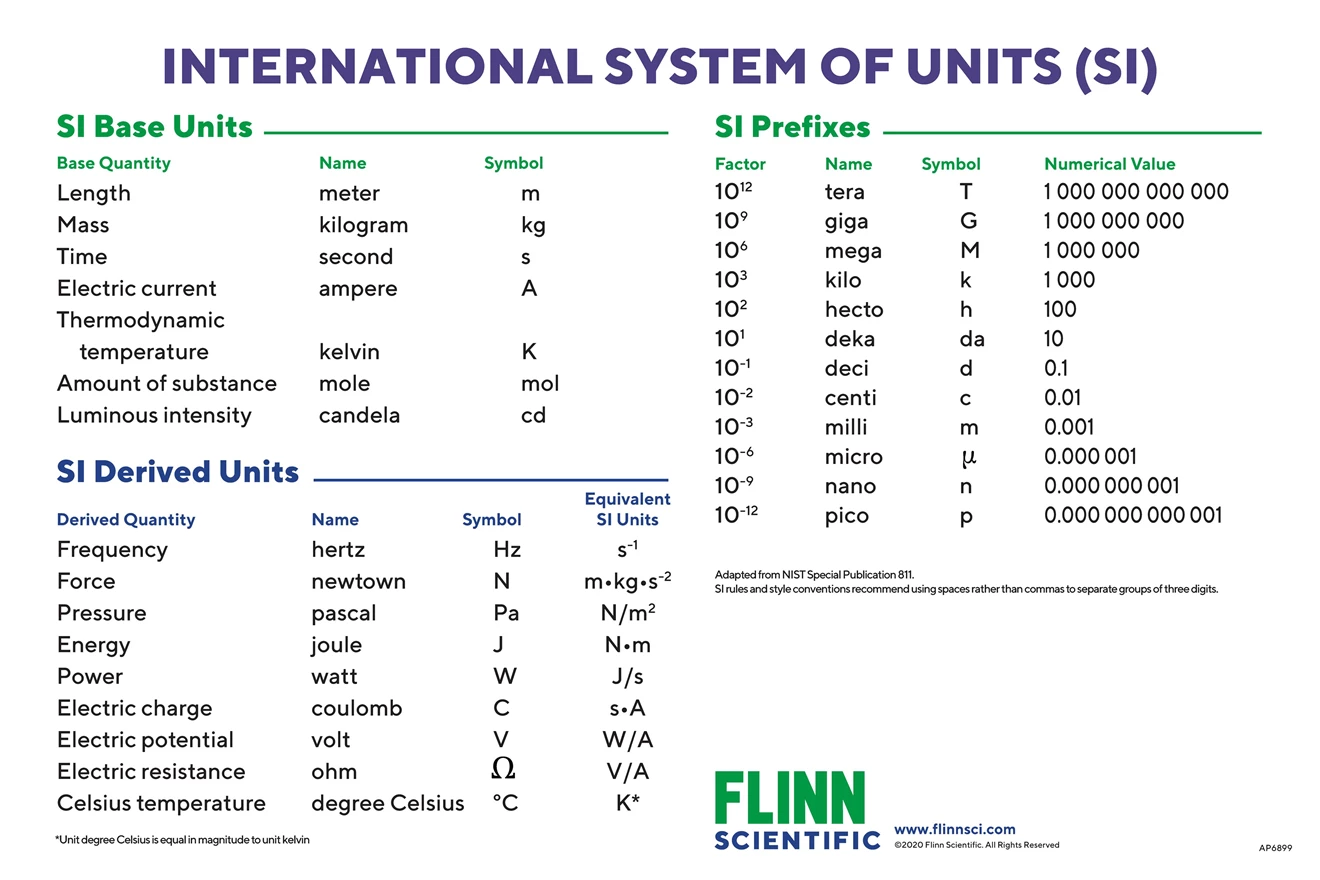
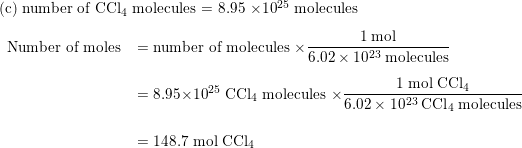SOLVED: Silicon tetrachloride (SiCl4) can be prepared by heating Si in chlorine gas: 2Cl2(g) + Si(s) → SiCl4(l) Calculate the number of moles of Cl2 required to form 0.178 moles of SiCl4.

SOLVED: Help me to determine Determine the mass, in grams, of 0.750 moles of Si (1 mol of Si has a mass of 28.09 g).